|
A newly-discovered enzyme may hold the key to longevity.
by Arthur Chee
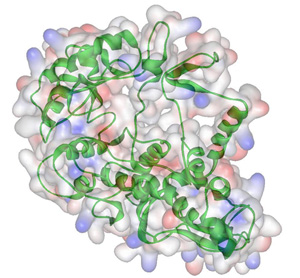 Photo courtesy
of Vimal Kapoor
Photo courtesy
of Vimal Kapoor
The predicted
3D structure of the PBEF enzyme.
|
|
On April 15th 2005, as part of the annual World-Wide Day in Science, I was lucky enough to spend a day with some researchers who are trying to determine whether a newly-discovered enzyme is a key factor in aging, and whether or not giving an organism more of it could slow down the aging process. Two years ago, pharmacology researcher Vimal Kapoor, then at the University of New South Wales in Sydney, Australia, was inspired by an article describing a newly-discovered enzyme. The researchers had identified this enzyme as Pre-B-cell colony Enhancing Factor or PBEF but were not sure of its exact role. The journal article was not widely distributed or cited but the results caught Kapoor's attention: he had a theory that this new enzyme might actually be a missing link in the chemical pathway that causes aging.
When we witness signs of aging, it's because a large amounts of cells in the body have reached the end of their useful lives and essentially disassemble themselves through a process called apoptosis. Cell death in areas like the heart, lungs, immune system, muscle, and nerve tissue occurs in old people and makes them physically weaker and prone to illness. When cells are young and healthy, a chemical called NAD helps provide cells with energy so that they can create new chemicals, and it is thought that PBEF, which is present in all of our cells, may play a significant role in the recycling of NAD. Supplying our cells with more PBEF could therefore be like giving them a new dose of energy: it could reduce the rate of programmed cell death and thus slow down the aging process.
Research into the role of PBEF has also linked it to a range of chemical mechanisms that regulate cell growth. Interestingly, some cancer cells express PBEF in larger quantities and may be part of the reason that they multiply so quickly. "Cancer cells live forever," says Kapoor, "and PBEF could be the key."
 Photo
courtesy of Arthur Chee
Photo
courtesy of Arthur Chee
Researcher
Piroska Bisits Bullen at work in the lab.
|
Kapoor and his research team, which
included neuroscience honours student Piroska Bisits Bullen, set
out to further investigate the role of this enzyme. Their first
challenge was to isolate PBEF and determine its DNA sequence. To
do this, they had to modify E. coli bacteria to produce the
enzyme artificially and then develop a process for purifying the
result so that they could isolate the enzyme. According to Bisits
Bullen, this step was the trickiest. "E.coli is very temperamental.
Working out exactly the right growing conditions to make them produce
the enzyme in the right way took over a year."
When I visited the lab, Bisits Bullen had successfully found numerous ways of producing the enzyme and after long hours of trial and error, the researchers had produced large quantities of the pure enzyme. Work was underway to test the effect of the enzyme on individual cells to see if it could really influence the rate of aging of organisms.
One year later, Kapoor and his research team have successfully produced crystals of the enzyme and are trying to determine its 3D structure using a technique called x-ray crystallography. "We will soon know what this enzyme looks like down to one or two Angstroms," says Kapoor. "From this, we hope to be able to develop novel anti-cancer drugs that can block it and thus slow the multiplication of cancer cells."
But they are still a long way off from extending our life span, which Bisits Bullen says poses many ethical issues. For example, at present the only way to deliver PBEF to all the cells in the human body would be to genetically modify a person's egg cell before it grows into a fetus, ensuring that the body's cell chemistry is altered from the moment that the person is conceived. It may be possible to develop different ways of delivering PBEF to cells, but this is still a major hurdle to overcome, and may take a decade or more.
In the fight against disease, however, one hope is that PBEF blockers could be used to initiate apoptosis in cancerous cells. Conversely, for other conditions that result from too much cell death, enhanced levels of PBEF levels could be used. Examples include Alzheimer's disease, which involves apoptosis of brain cells, sepsis due to a degrading of the immune system, and forms of diabetes, where insulin-producing cells have died out.
Eternal youth? Perhaps one day, but it seems that PBEF may initially be destined to prolong the lives of those who need it most.
Participate in this year's World-Wide Day in Science on April 12th 2006:
You can take part this year if you
are interested in science, as a student, scientist, teacher, or
other science-based professional. Tell us the high points of your
"day in science" on April 12th (even if you read this article after April
12th).
To find out how to participate, please visit:
http://www.science.unsw.edu.au/worldwide/
You can also read more about Arthur Chee's experience last year at:
http://au.geocities.com/arthur_wwds/piroska.htm
|
